|
11/11/2022 0 Comments Bond angle of sf2 Related questions How do I determine the bond angle in a molecule? Question b Question 2a64e. Apr 21, The tellurium atom will also have one lone pair of electrons attached. However, repuslion will cause the lone pair of electrons present on the tellurium to distort these angles a bit by pushing the bonding electrons away from it. 
The central atom will have a steric number equal to 5since the tellurium atom is surrounded by five regions of electron density - four single bonds and one lone pair of electrons. The tellurium atom will the the central atom of the molecule, the four chlorine atoms being bonded to it through single bonds. The molecule has a total of 34 valence electrons6 from the tellurium atom and 7 from each of the four chlorine atoms. You can determine its molecular geometry by drawing its Lewis structure. Still have questions? Get your answers by asking now.You can predict the bond angles of tellurium tetrachloride by looking at its molecular geometry. Fauci: Trump ad takes my words out of context.Īnswer Save. Wallace grills Lara Trump for breaking debate rule. The undocumented Latina leading a massive movement. How 'secretly in love' bandmates finally came out. Beware of appropriation posing as a costume. Trump turns power of state against his political rivals. Ex-Obama adviser: Covid infections 'going to go up'. 
Hailey Bieber endorses Biden - while dad backs Trump. Therefore the shape of BCl3 is trigonal planar and the bond angle is degree. Hence the bond angle is even smaller than The approximate bond angle is about For BCl3, the boron atom is surrounded by three electron pairs and the three electron pairs are all bonding pairs. The repulsive force between lone pair - lone pair is greater than between the lone pair - bond pair. 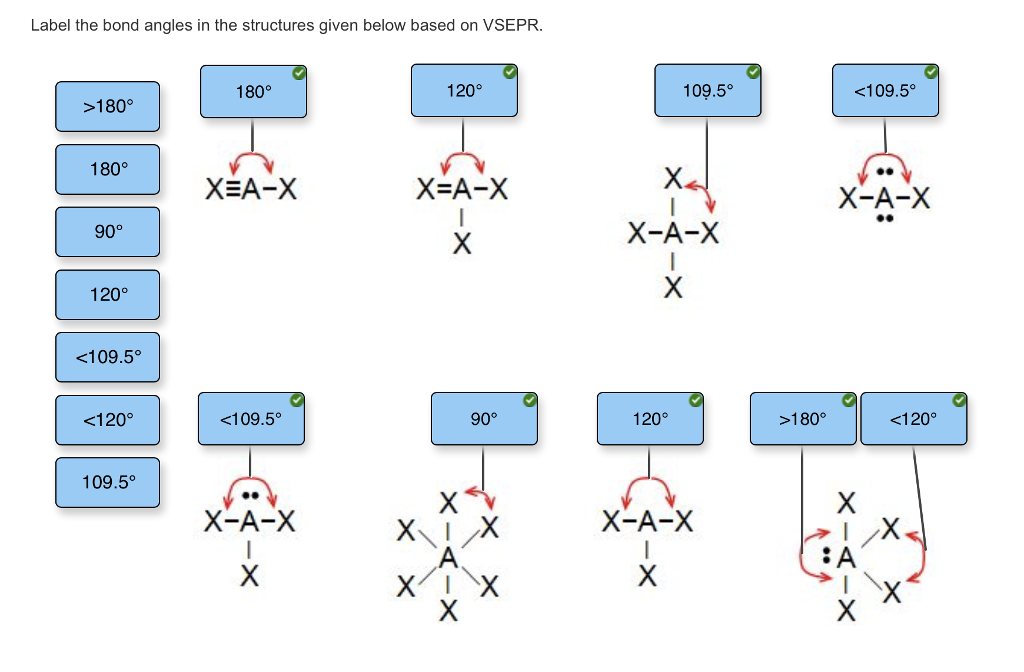
For SF2, the sulphur atom is surrounded by four electron pairs but there are two bonding pairs and two lone pairs. Hence the bond angle is slightly smaller than The approximate bond angle is about degree. The repulsive force between lone pair - bond pair is greater than the bond pair - bond pair. Therefore, the shape of NI3 is trigonal pyramidal. Therefore they have the tetrahedral shapes with the approximate bond angles of For NI3, the nitrogen atom is surrounded by four electron pairs but there are three bonding pairs and one lone pair. Subscribe to: Post Comments Atom.The carbon atom is surrounded by four electron pairs and all the electron pairs are the bonding pairs. 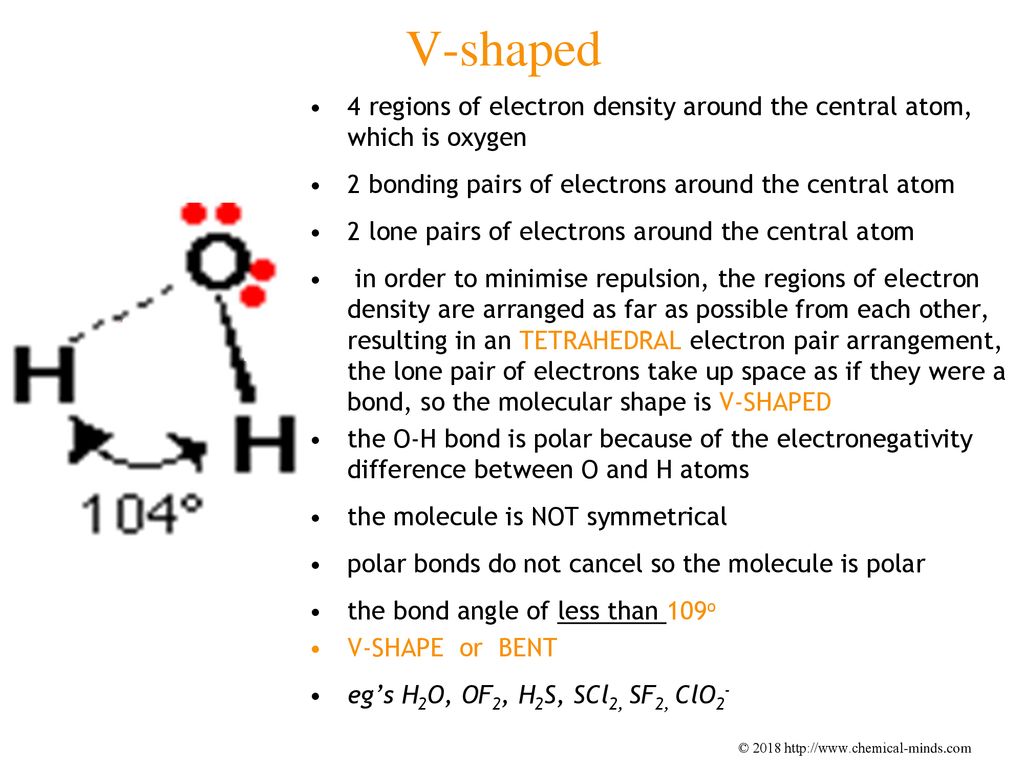
Monday, 17 September What are the approximate bond angles? The carbon atom is surrounded by four electron pairs and all the electron pairs are the bonding pairs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
